Our primary work is with medical device and IVD manufacturers — companies entering or operating in the UK and EU markets who need proper regulatory representation. We also work with industrial and commercial energy users, and with manufacturers requiring product conformity support.
Home » Sectors & Capability
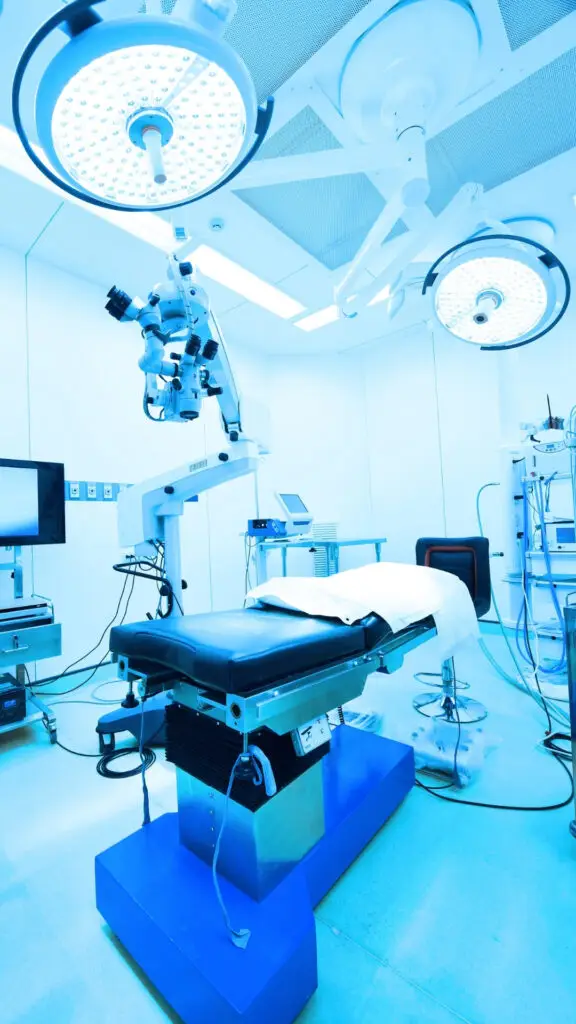
Our primary area of activity. We work with medical device and IVD manufacturers at every stage of market entry and ongoing regulatory compliance — from initial UKRP or EC-REP appointment through to MHRA registration, EUDAMED management, and post-market surveillance support.
Manufacturers in this sector face a regulatory environment that continues to evolve. MDR and IVDR transition timelines, EUDAMED obligations, and MHRA post-Brexit requirements all require active management — not a one-time submission. We provide that ongoing management as a registered representative, not as an advisor standing at a distance.

Commercial and industrial energy users qualifying under ESOS or the UK Energy Regulations 2024 have defined compliance obligations and, in many cases, significant potential for energy cost reduction and environmental performance improvement.
We support organisations through the compliance process and, where capital improvement projects are required, co-ordinate with specialist mechanical and electrical partners to deliver under an EPCC structure. Energy Managers, Environmental Managers, Sustainability Leads, and Engineering and Procurement teams are our typical points of contact in this sector.

Manufacturers and production facilities in this sector typically require a combination of management system support, product conformity assessment, and supply chain assurance. We deliver audit programmes, machinery and pressure equipment inspection, and CE/UKCA marking support for operators who need independent, technically credible third-party delivery.
Our background in manufacturing governance, calibration environments, and production QC means findings are grounded in operational reality — not theoretical compliance frameworks.

Construction and contracting environments combine occupational health and safety obligations, product conformity requirements for plant and equipment, and supply chain compliance management — often across multiple sites and subcontractors simultaneously.
We provide ISO 45001 audit support, machinery conformity assessment, LOLER and PSSR inspection co-ordination, and subcontractor audit programmes for principal contractors and specialist firms who need credible, independent delivery without the cost of a large consultancy engagement.

Our primary area of activity. We work with medical device and IVD manufacturers at every stage of market entry and ongoing regulatory compliance — from initial UKRP or EC-REP appointment through to MHRA registration, EUDAMED management, and post-market surveillance support.
Manufacturers in this sector face a regulatory environment that continues to evolve. MDR and IVDR transition timelines, EUDAMED obligations, and MHRA post-Brexit requirements all require active management — not a one-time submission. We provide that ongoing management as a registered representative, not as an advisor standing at a distance.
Discover how we’ve helped organizations solve complex challenges and achieve their objectives.
Faced with stringent regulations, our client needed to align their operations with compliance standards. Through thorough assessments and tailored solutions, we enabled them to enhance their processes and reduce risks, achieving a 30% increase in compliance scores.

Through strategic energy assessments, we assisted a leading firm in identifying inefficiencies in their energy use. As a result, they implemented our recommendations and decreased their energy costs by 25%, contributing to their sustainability goals.
Have questions? We’re here to help clarify your needs and guide you through our services. Let’s connect to explore how we can assist you.
By submitting, you agree to our Terms and Conditions.